A team of researchers from IBM Research Europe, Universidade de Santiago de Compostela, and the University of Regensburg has changed the bonds between atoms for the first time. The group describes their method in a paper that was published in Science. A piece about the work done by the team has been published in the same journal issue.
The current method for creating complex molecules or devices is difficult, like dumping a box of Legos in a washing machine and hoping that some useful connections are made, as Alagugin and Chaowei note. The research team has made it easier to break bonds in a molecule by using a scanning tunneling microscope (STM) and then modifying the molecule by creating new bonds.
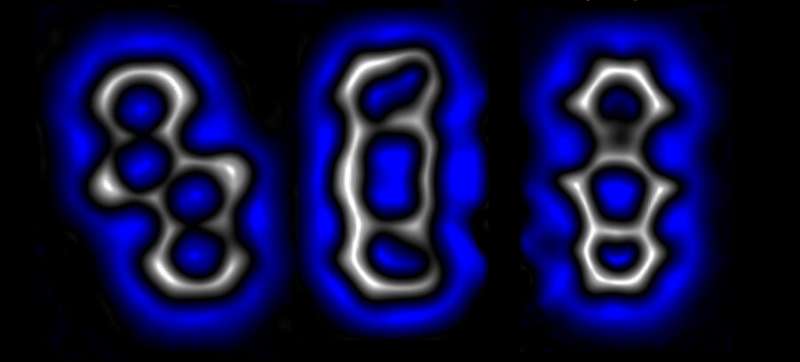
The team used a very small amount of electricity to break bonds in a sample material that was placed into a scanning tunneling microscope. The four chlorine atoms were pulled from the core of a tetracyclic to use as their starting molecule.
They broke the C-CI bond with electricity after moving the tip of the STM to a C-CI bond. Doing so to the other C-CI and C-C pairs resulted in the formation of a diradical, which left six electrons free to be used in forming other bonds.
In one test of creating a new molecule, the team used the free electrons to form diagonal C-C bonds, which resulted in the creation of a bent alkyne. The cyclobutadiene ring was created when they applied a dose of low voltage.
According to the researchers, their work was made possible by the development of ultrahigh precision tunneling technology, which was developed by a team headed by Gerd Binnig and Heinrich Rohrer, both with IBM’s laboratory in Zurich. They suggest that their technique could be used to create new types of molecules.
Further information
Florian Albrecht et al, Selectivity in single-molecule reactions by tip-induced redox chemistry, Science (2022). DOI: 10.1126/science.abo6471
Igor Alabugin et al, A Swiss Army knife for surface chemistry, Science (2022). DOI: 10.1126/science.abq2622
Latest Chemistry Research News
- Scientists developed durable material for flexible artificial muscles
- Solar-powered chemistry uses carbon dioxide and water to make feedstock for fuels, chemicals
- Team of scientists creates first ever VX neurotoxin detector
- Scientists Discovered working principles of a promising material for fast-charging batteries